Purpose:
The purpose of this lab is to see if plants collected in our field study sites contain active ingredients that could inhibit the growth of bacteria.
Materials:
|
Balance, weigh boat, lab scoops
LB broth base 250 mL Media bottles Sterilizer/autoclave 37 degree Celsius Water Bath Sterile LB Agar Laminar flow hood and disinfectant Safety Goggles Bunsen burner and gas lighter |
Inoculating loop, Ni/Cr wire
Sterile Petri dishes, 60x15mm E. coli JM109 Plant from field study site Mortar and pestle 10 mL pipette Plastic funnels 5 mm diameter Filter Paper disks Beakers, 100 mL |
Syringe, 10 mL and filter
Reaction tubes and rack, 1.7 mL Methanol, absolute 1 mL Pipette Heat Block Fine-tipped Forceps Ampicillin Glass Spreader 37 degrees Celsius Incubator Oven |
PROCEDURE:
Part II:
- Grind 2g of plant using mortar and pestle with 10 mL of de-ionized water.
- Let sit or 3 minutes
- Filter through a paper-lined funnel
- Sterilize using a filter and put into a 1 mL microtube
- Repeat using 10 mL Methanol
- Put tube in heat block with lid open and leave overnight (evaporation)
- Using flame-sterilized forceps, insert 2 discs in each tube
- Repeat with a positive (Ampicillin) and negative (Water) control tube
- Leave overnight at 4 degrees Celsius
Part III
- Use sterile pipette to transfer 1 mL of E. coli culture to petri disc (divided in 4 quadrants)
- Spread with flame sterilized glass spreader around disc
- Cover and let soak for 15 minutes
- Use sterile forceps to place discs on plates in each quadrant
- Incubate at 37 degrees Celsius for 24 hours
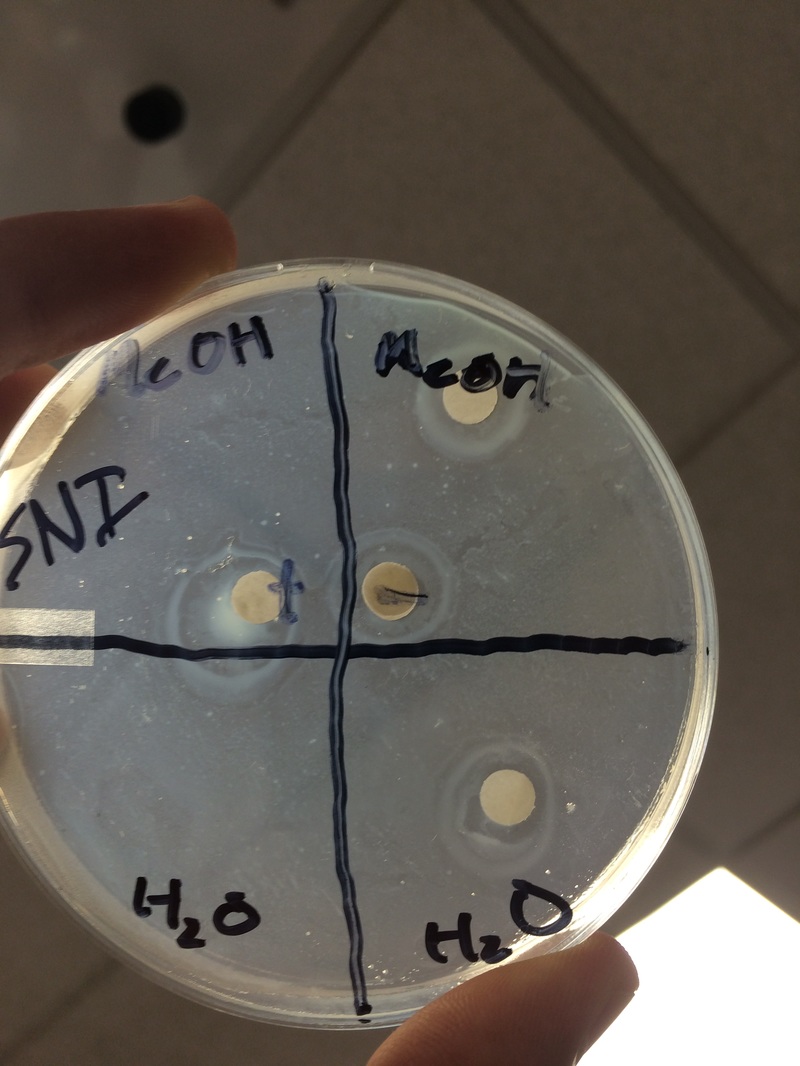
Results:
- My positive control turned out as expected; a 1.5 mm diameter circle of bacteria-free area was created around the disk soaked in Ampicillin.
- My negative control also turned out as expected as a 1 mm diameter circle was created full of bacteria around the water-soaked disk.
- Due to difficulties with the removal of our extract disks, we were only able to have one of each extract on the disk, however both my partner and my experiments turned out the same. Our Methanol disk was surrounded by a 1 mm diameter circle covered with bacteria while our water disk had a 1 mm diameter circle without any bacteria present.
Analysis/Conclusion:
My water extract of my site plant yielded positive results as evident by the 1 mm circle surrounding the disk that was bacteria-free. My controls both worked as expected with my positive control inhibiting bacteria growth and my negative control allowing free growth of bacteria. While I didn't have any major errors, sources of some errors could be contamination due to improper sterilization, an incompletely ground plant extract, or spilling some liquid on the bacteria dish. To improve this experiment, I would use more disks to have a better chance at using two disks for each extract. This would make our results much more accurate and reliable. Some next steps to take could be to attempt the experiment again with different flowers from the same plant to make sure our results are accurate as well as possibly testing other parts of the plant for anti-bacterial properties.
Thinking Like a Biotechnician Questions:
- If an extract gives a negative result in the antimicrobial assay, it is possible that the extract is still an antimicrobial agent, but just not for that particular bacteria. Further tests could determine if the extract has any antimicrobial properties.
- In preparing the sample disks, some of the methanol extractions smelled like alcohol. This could be a problem because the presence of alcohol could affect the bacterial growth.
- To identify the exact compound in an extract that causes the antimicrobial action in our plant extracts, we could separate individual components using chemical reactions and then test each of them to figure out which causes the reaction. It is possible that when multiple parts are needed for the effect, so you may need to try combinations of the separated components.